Research Topics
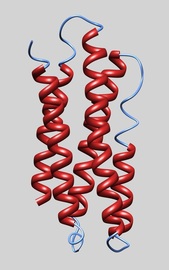
Apolipoproteins are the protein components of lipoproteins, and their role in lipid transport processes has been well documented. It is much less known that apolipoproteins also play an important role in innate immunity and our research program aims to understand this novel function. To study these critical players in lipid transport processes and innate immunity, we use two different apolipoproteins: human apolipoprotein A-I (apoA-I, 28 kDa) and insect apolipophorin III (apoLp-III, 18 kDa). ApoA-I is an anti-atherogenic protein and the main protein component of high-density lipoproteins, which are known for reverse cholesterol transport. ApoLp-III has proven to be an excellent model system and has the advantage of known three-dimensional structures, which is critical to our structure-guided approach. Through a structure-guided site-directed mutagenesis approach we aim to obtain fundamental knowledge of how apolipoproteins function at the molecular level, gaining critical knowledge of their structure-function relationship. This allows us to understand the molecular basis of diseases associated with apolipoprotein function.
ApoLp-III and apoA-I. We investigate the antimicrobial properties of apoLp-III and apoA-I and look at the lipopolysaccharide and phosphatidylglycerol binding interaction (both abundantly present in membranes of gram-negative bacteria). Oztug et al., 2012, Biochemistry 51, 6220-6227; Beck et al., 2013, Biochim. Biophys. Acta 1828, 1503-1510.
ApoLp-III structure. We are interested in how the helix bundle structure facilitates lipid binding. A site-directed mutagenesis approach is employed, using X-ray and NMR solution structures of apoLp-III as a guide. Dwivedi et al., 2016, Biochemistry 55, 3607-3615.
The C-terminal domain of apoA-I. The C-terminal domain of apoA-I is a relatively unstructured part of the protein, consisting of two putative helices. The C-terminal helices are responsible for the initiation of lipid binding, leading to the uptake of phospholipids and formation of discoidal lipoproteins. Residues in the C-terminal domain also mediate apoA-I self-association. We are carrying out a mutagenesis study to identify the specific amino acid residues for these processes. Fuentes et al., 2018, Biochemistry 57, 2200-2210.
Chimera proteins. We are designing chimera proteins using apoLp-III as a carrier to attach domains or segments of other apolipoproteins. Recently attached the C-terminal domain of apoA-I to apoLp-III, which acquired apoA-I like properties (high lipid binding and self-association). We are in the process of generating additional chimeras to gain insight in the domain organization of apolipoproteins. Horn et al., 2017 Biochim. Biophys. Acta, 1859, 1317-1325; Lek et al., 2017, PLoS One 12(6):e0178346.
ApoLp-III and apoA-I. We investigate the antimicrobial properties of apoLp-III and apoA-I and look at the lipopolysaccharide and phosphatidylglycerol binding interaction (both abundantly present in membranes of gram-negative bacteria). Oztug et al., 2012, Biochemistry 51, 6220-6227; Beck et al., 2013, Biochim. Biophys. Acta 1828, 1503-1510.
ApoLp-III structure. We are interested in how the helix bundle structure facilitates lipid binding. A site-directed mutagenesis approach is employed, using X-ray and NMR solution structures of apoLp-III as a guide. Dwivedi et al., 2016, Biochemistry 55, 3607-3615.
The C-terminal domain of apoA-I. The C-terminal domain of apoA-I is a relatively unstructured part of the protein, consisting of two putative helices. The C-terminal helices are responsible for the initiation of lipid binding, leading to the uptake of phospholipids and formation of discoidal lipoproteins. Residues in the C-terminal domain also mediate apoA-I self-association. We are carrying out a mutagenesis study to identify the specific amino acid residues for these processes. Fuentes et al., 2018, Biochemistry 57, 2200-2210.
Chimera proteins. We are designing chimera proteins using apoLp-III as a carrier to attach domains or segments of other apolipoproteins. Recently attached the C-terminal domain of apoA-I to apoLp-III, which acquired apoA-I like properties (high lipid binding and self-association). We are in the process of generating additional chimeras to gain insight in the domain organization of apolipoproteins. Horn et al., 2017 Biochim. Biophys. Acta, 1859, 1317-1325; Lek et al., 2017, PLoS One 12(6):e0178346.
